AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Iso 22716 audit checklist pdf8/29/2023  PLI/PAIs cover cGMPs, but also – Verify information submitted for review tuvi saq.ウェブ– Routine GMP inspections – quality systems, adherence to cGMPs, follow-up from previous inspections (may be full, i.e., all six systems or abbreviated) ĮDQM inspections and trends of GMP deficiencies: Overview. Preparing for the Pre-Approval Inspection What to do Before. What is GMP Good Manufacturing Practices Safet圜ulture GMP Auditing for Quality Assurance Training Course CAPT United States Public Health Service Supervisory. 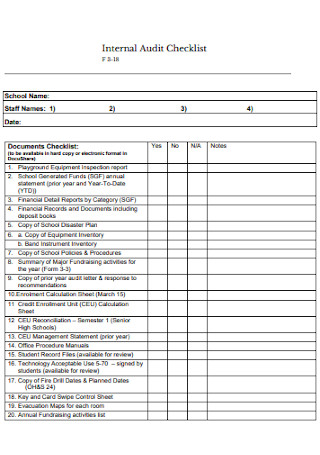
ウェブApproaches to GMP inspection CDER Small Business - Regulatory Education for Industry (REdI) June 2014 Uduak M. ISO 22716:2007 Auditor Training - Editable PPT Presentation. This is troubling because ensuring data integrity is an important component of. 
ウェブIn recent years, FDA has increasingly observed CGMP violations involving data integrity during CGMP inspections. 
FDA has developed training courses, webinars, and other teaching materials that are designed to help foreign and domestic industry, medical professionals, regulators, academics and others better. 
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
